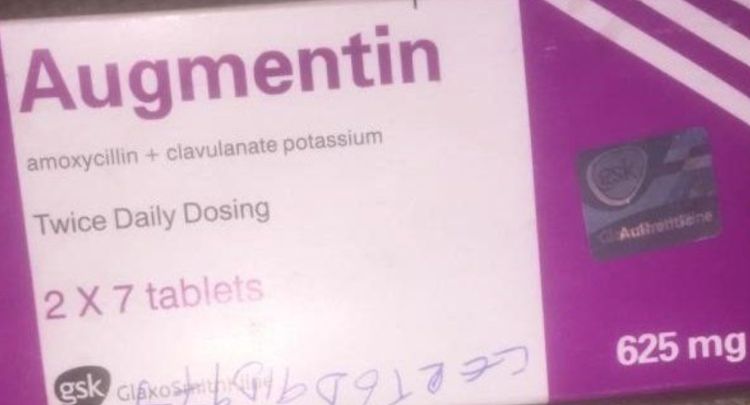
The National Agency for Food and Drug Administration and Control (NAFDAC) has warned Nigerians about counterfeit Augmentin 625mg tablets reportedly circulating in the country.
In a statement issued Friday, the agency said the alert followed complaints received by GlaxoSmithKline (GSK), the original manufacturer of Augmentin, concerning suspected fake versions of the medication in circulation.
According to NAFDAC, GSK confirmed the product was counterfeit after examining “documentary evidence, photographs, and a video from social media.”
“Although the product shared the same batch number as authentic stock previously supplied to Nigeria, it exhibited inconsistent manufacturing and expiry dates, indicating that it had been falsified,” the statement said.
Providing details from the manufacturer’s investigation, NAFDAC explained that batch AC3N was originally produced in Worthing, United Kingdom, on August 23, 2023, and packaged between September 19 and 20, 2023.
The agency added that “no batch with the number AC3N was produced in September 2025,” noting that the manufacturing and expiry dates found on the suspected counterfeit products — September 2025 and September 2028 — “are inconsistent with GSK production records and cannot be attributed to a GSK-produced batch.”
NAFDAC also identified “packaging-level discrepancies, including textual errors and poor fin-seal quality.”
The agency warned that counterfeit medicines pose serious risks to public health.
“The use of counterfeit Augmentin 625mg tablets may result in treatment failure, worsening of bacterial infections, development of antimicrobial resistance, adverse drug reactions due to unknown or harmful ingredients, and delay in receiving appropriate medical care,” it said.
Augmentin, which contains amoxicillin and clavulanic acid, is widely prescribed for the treatment of bacterial infections.
NAFDAC said all zonal directors and state coordinators had been instructed to strengthen surveillance efforts and remove counterfeit products from circulation nationwide.
The agency advised importers, distributors, retailers, healthcare professionals, and consumers to purchase medicines only from authorised suppliers and to carefully inspect products before use.
“Consumers who have purchased or are currently using Augmentin 625mg tablets suspected to be counterfeit should stop using the product immediately and consult a qualified healthcare professional,” the statement added.